Kjar, A.; Haschert, M.R.; Zepeda, J.C.; Simmons, A.J.; Yates, A.; Chavarria, D.; Fernandez, M.; Robertson, G.; Abdulrahman, A.M.; Kim, H.; Marguerite, N.T.; Moen, R.K.; Drake, L.E.; Curry, C.W.; O’Grady, B.J.; Gama, V.; Lau, K.S.; Grueter, B.; Brunger, J.M.; Lippmann, E.S. Cell Reports, Volume 43, Issue 11, 2024, Article 114874, Ь§
Ь§
Human neural organoids are used to study brain biology, but making them more accurate in representing different types of brain cells is still a challenge. In this study, the researchers compared two materials for growing these organoids: Matrigel (a common material) and a new material called GelMA-Cad, which is made from a special gel that helps guide cell development. They found that the GelMA-Cad material helped the organoids develop more like human fetal brain tissue and produced neurons that were more active compared to those grown in Matrigel.Ь§
These findings suggest that GelMA-Cad could be a better material for growing neural organoids, allowing researchers to control how the cells develop more precisely. It also offers a simpler and more reliable alternative to Matrigel for research on brain development.Ь§
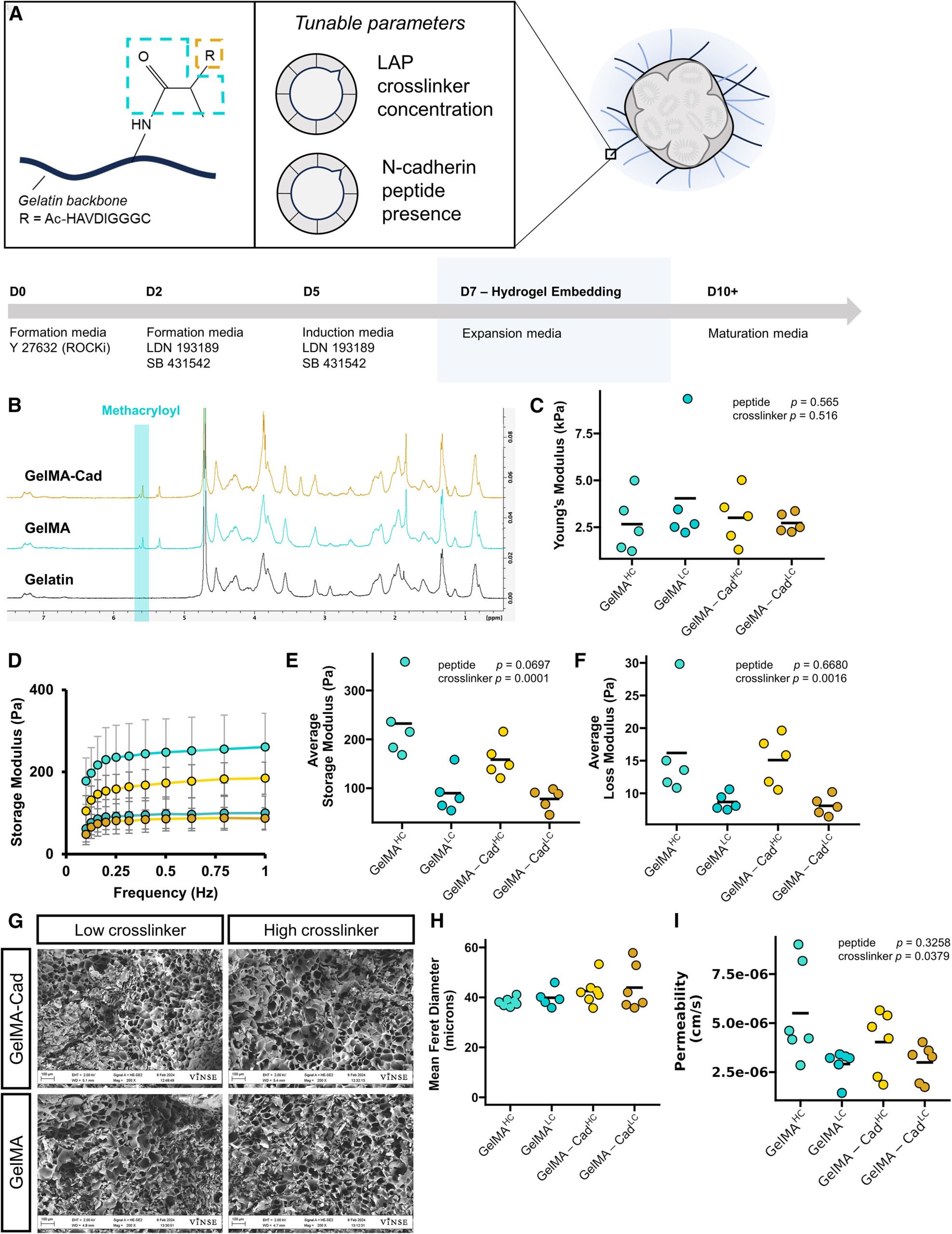
Figureܧ1.ܧFunctionalized gelatin matrix parameters for cortical organoid cultureܧ
(A) Schematic of GelMA-Cad, major tunable parameters, and associated organoid culture timeline. GelMA-Cad utilizes a gelatin-based backbone, with a conjugated methacryloyl group (blue), allowing photo-initiated crosslinking. The methacryloyl can be further modified by the addition of an N-cadherin (Cad) peptide mimetic (orange). Adjusted parameters include the LAP crosslinker concentration and N-cadherin peptide presence.Ь§
(B) Representativeܧ1H-NMR spectra of gelatin, GelMA, and GelMA-Cad displaying the characteristic peaks associated with methacryloyl.ܧ
(C) Atomic force microscope characterization of Youngтs modulus.Ь§NЬ§= 5 measurements, each the average of 512 technical replicates. Bars represent data mean. Statistical significance was calculated with a two-way ANOVA, modeled on crosslinker concentration and peptide presence.Ь§
(D) Average storage modulus traces from GelMA and GelMA-Cad hydrogels.ܧNܧ= 5 measurements, error bars represent the standard deviation.ܧ
(E) Rheological characterization of average storage modulus across tested frequencies.ܧNܧ= 5 measurements, bar represents data mean. Statistical significance was calculated with a two-way ANOVA, modeled on crosslinker concentration and peptide presence.ܧ
(F) Rheological characterization of average loss modulus across tested frequencies.ܧNܧ= 5 measurements, bar represents data mean. Statistical significance was calculated with a two-way ANOVA, modeled on crosslinker concentration and peptide presence.ܧ
(G) Representative scanning electron micrographs of lyophilized, crosslinked hydrogels. Scale bar: 100Ь§ЮМm.Ь§
(H) Quantification of pore size from scanning electron micrographs. Each point is the mean of measurements made from a single image,Ь§NЬ§= 6т7 images per condition, each image from a separate preparation. Bars represent data mean.Ь§
(I) Apparent permeability coefficient of 3ܧkDa FITC-dextran in each hydrogel.ܧNܧ= 6 measurements, bar represents data mean. Statistical significance was calculated with a two-way ANOVA, modeled on crosslinker concentration and peptide presence.ܧ